The Function and Molecular Mechanism of Cellular Senescence-Inhibited Gene
Liwei Ma, Feng Zhu, Nan Xie, Feng Tian, Ying Sun and Tanjun Tong
DOI10.21767/2573-5365.100016
Liwei Ma, Feng Zhu, Nan Xie, Feng Tian, Ying Sun and Tanjun Tong*
Research Center on Aging, Department of Biochemistry and Molecular Biology, Peking University Health Science Center, Beijing, 100191, P.R. China
- Corresponding Author:
- Tanjun Tong
Department of Biochemistry and Molecular Biology,Peking University Health Science Center,38 Xueyuan,Road, Beijing 100191, P.R. China
Tel: + 8610-82801454
Fax: + 8610-82802931
E-mail: tztong@bjmu.edu.cn
Received date: May 06, 2016; Accepted date: May 27, 2016; Published date: May 29, 2016
Citation: Ma L, Zhu F, Xie N, et al. The Function and Molecular Mechanism of CSIG. Cell Mol Med. 2016, 2:2.
Copyright: © 2016 Ma L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
To explore the genetic mechanism of replicative senescence, we identified and cloned a Cellular Senescence-Inhibited Gene/ribosomal L1 domain containing 1 (CSIG/RSL1D1) through suppressive subtractive hybridization. CSIG expression is high in earlypassaged human diploid fibroblast cells but decreased during senescence. Our results demonstrated that CSIG inhibited the senescence progression and mediated the apoptosis signaling pathway under ultraviolet (UV) irradiation. Moreover, emerging evidences have indicated that CSIG is involved in other processes including cell cycle regulation and tumor development. This is a brief overview to introduce the studies on the function and molecular mechanism of CSIG
https://transplanthair.istanbul
https://hairclinicturkey.co
https://hairclinicistanbul.co
https://besthairtransplant.co
https://hairtransplantistanbul.co
Introduction
Replicative senescence, a cellular self-protection mechanism to prevent tumor development, contributes to the generation of age-related disorders [1-3]. As the expression alteration of important genes is the molecular basis of replicative senescence, it is necessary to explore the gene expression and signal transduction mechanism during senescence. By analyzing the gene expression profiles in earlypassaged and senescent fibroblasts, Guo et al. cloned and characterized Cellular Senescence-Inhibited Gene [CSIG, GenBank accession No. AY154473, also named as ribosomal L1 domain containing 1 (RSL1D1) according to https://www.ncbi.nim.nih.gov] through suppression subtractive hybridization in human diploid fibroblasts [4]. CSIG expression is high in early-passaged human diploid fibroblast cells but decreased during senescence [4,5]. Tsai et al. reported that the decreased proliferation of aging tenocytes is associated with the down-regulation of CSIG [6]. The experiments in vivo and in vitro both indicated that CSIG is not only associated with cellular senescence but also participates in organismal aging [4]. And our previous work proved that CSIG delays cellular senescence by inhibiting the phosphatase and tensin homolog (PTEN)/p27Kip1 pathway [5].
In addition to the cellular senescence, CSIG is also involved in many other important biological processes. For example, under ultraviolet (UV) irradiation, CSIG mediates the apoptosis signaling pathway [7]. In the process of bone healing and growth, CSIG expression is increased [8], indicating the participation of CSIG in the bone tissue formation, reabsorption, regeneration and healing. Inflammations such as respiratory tract inflammation are accompanied with increased CSIG gene expression [9]. Cheng et al. reported that CSIG facilitated the proliferation of hepatocellular carcinoma [10]. By analysis of dog osteosarcoma samples, Selvarajah et al. found that CSIG was not only related to tumor cells survival but also associated with cell proliferation and cancer metastasis [11], and therefore CSIG may act as a new therapeutic target. Furthermore, Klein et al. found that CSIG expression in breast cancer brain metastases samples was significantly higher than in bone metastases [12]. Thus CSIG may be used as a predictive marker for specific tissue metastasis of breast cancer and offers the possibility of personalized treatment. In addition, Fujiyama-Nakamura et al. found that CSIG was involved in the ribosomal protein synthesis. They detected the interaction of CSIG with Parvulin, a peptidyl-prolyl cis-trans isomerase, in the process of ribosomal protein synthesis [13].
By microarray analysis, we recently discovered that the downstream genes modulated by CSIG are mainly implicated in the processes of cell cycle, apoptosis, transcriptional control, bone formation and cell adhesion [14], which further provides the molecular basis for CSIG to participate in these biological processes.
Identification and characteristics of CSIG
By comparing the expression profiles of early-passaged human fibroblast cells with that of senescent cells, we previously identified CSIG/RSL1D1 through suppressive subtractive hybridization method [4]. CSIG is abundant in early-passaged fibroblast cells while declines during senescence [4,5]. By localizing on chromosome 16p13m, the human CSIG gene spans 5,137 bp, contains 9 exons and encodes 490 amino acids residues. According to bioinformatics analysis, CSIG encodes a short half-life alkaline protein with the molecular weight of 54.7 kDa and the isoelectric point (PI) of 10.09. We observed that CSIG was widely expressed in human most tissues with higher expression in heart, skeletal muscle and placental tissues [5]. And the immunofluorescence analysis showed that CSIG protein was predominantly distributed in the nucleolus [5].
As a member of the ribosomal protein L1p family, CSIG is evolutionarily conserved according to the analysis of bioinformatics (https://www.expasy.org). There is a ribosomal protein L1-like domain (residues 31-264) in the CSIG protein Nterminus, indicating that CSIG may be involved in ribosome biosynthesis. As one of the largest ribosomal proteins, ribosomal L1P from the archaea binds directly to 23S rRNA. The L1 stalk is quite mobile in the ribosome, and it is involved in E site tRNA release. In bacteria and archaea, L1 acts as an rRNA-binding ribosomal protein and a translation repressor by binding its own mRNA [15]. The RNA-binding site of L1 is highly conserved, with both mRNA and rRNA binding the same binding site [16]. Like several other large ribosomal subunit proteins, L1 displays RNA chaperone activity [17]. Human CSIG protein C-terminus consists of a long lysine (Lys)-rich region (residues 282-485) similar to many ribosomal proteins including L14, L21, L22, L23, L24, L29 and L35. Our previous results proved that CSIG C-terminal Lys-rich region interacted with the inhibitor of growth family member 1 (ING1, also as p33ING1) [6]. Meng et al. reported that the C-terminal (residues 317-452) part of mouse homolog RSL1D1 without the L1 domain bound to nucleostemin [18]. These evidences suggested a role of protein binding activity for CSIG Cterminus.
CSIG and senescence
As described above, CSIG is abundant in early-passaged fibroblast cells, but declines in senescent cells. Consistently, in human peripheral blood leukocytes, we observed the same expression pattern of CSIG with age (CSIG level is significantly higher in the samples from the newborns while lower in that from old peoples) [4], which suggested that CSIG might be implicated in aging in vitro and in vivo. Coincidentally, Chiocchetti et al. reported that yeast protein RPL10, which shares a similar domain with CSIG, could affect the life span of yeast [19].
The human diploid fibroblast cells are generally served as the established model of replicative senescence. Just using this model, we firstly explore the function of CSIG during senescence and the possible mechanism. Through functional studies we found that CSIG could significantly affect the cell cycle and inhibit the cellular senescence progression [5]. CSIG overexpression increased the percentage of S phase cells, promoted cell proliferation, reduced the senescenceassociated β-galactosidase (SA-β-gal) activity, and prolonged the cell lifespan by more than 10 population doublings (PDs) [5]. Conversely, CSIG silencing increased the percentage of G1 phase cells, inhibited cell growth, increased SA-β-gal activity, and shortened the cell lifespan by accelerating cell entry into the stable G1 phase arrest status (named as senescence) [5].
The results suggested that the high level of CSIG contributed to maintain the active state of early-passage cells, while the decline of CSIG expression during senescence progression might be well causally implicated in generating senescence phenotype.
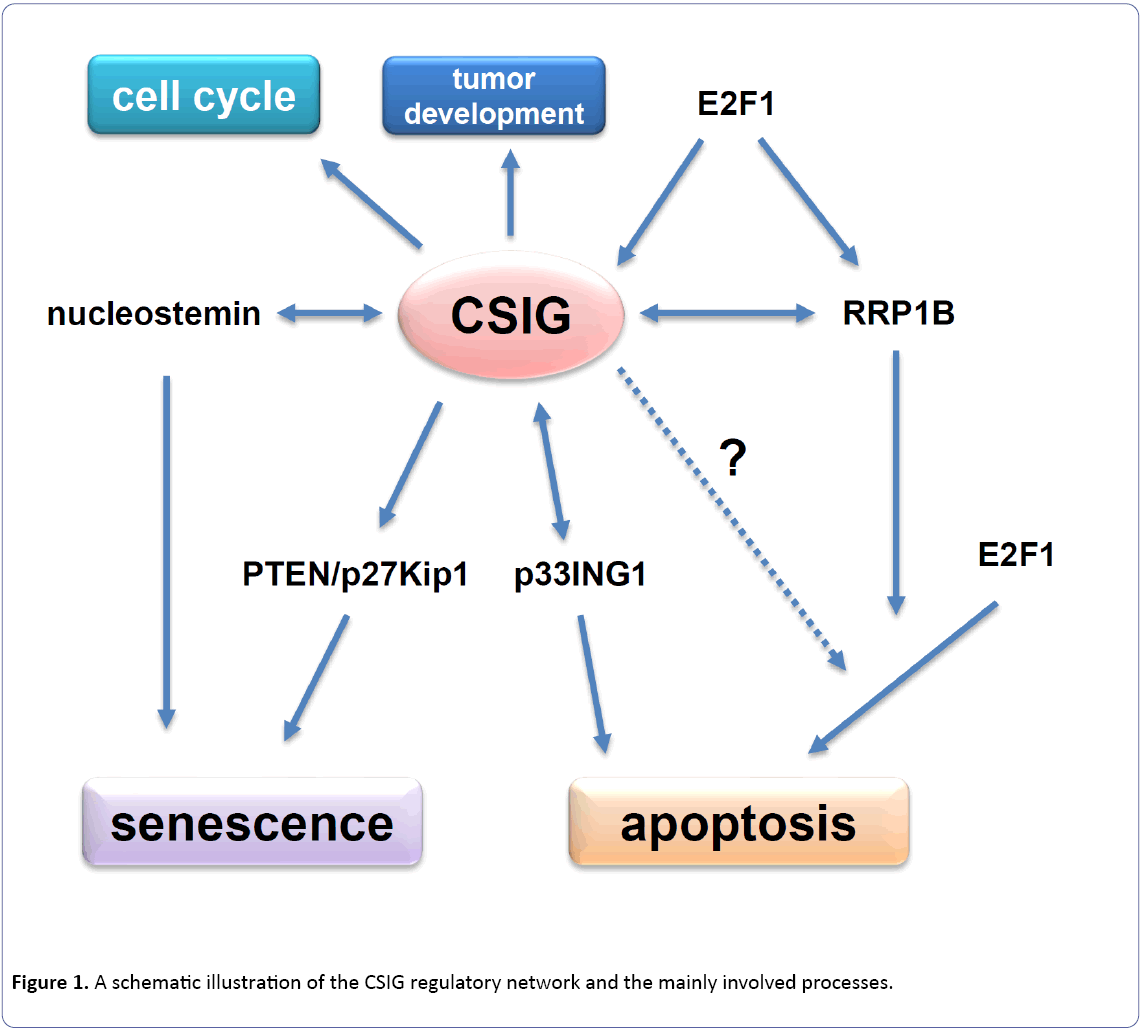
Our further study showed that CSIG delayed cellular senescence through down-regulating PTEN/p27Kip1 pathway [5]. During senescence, CSIG expression declines and its inhibition on tumor suppressor gene PTEN and cyclindependent kinase inhibitor p27Kip1 diminishes accordingly. Then the accumulated PTEN in turn leads to the cell cycle G1 phase arrest through increasing the level of p27Kip1. Our results also indicated that CSIG could inhibit the synthesis of PTEN nascent protein and PTEN was a necessary factor for CSIG to modulate cellular senescence [5]. Totally, our findings suggest that CSIG plays an inhibitory role during cellular senescence and PTEN acts as an essential mediator for CSIG to regulate senescence [5]. The cell aging-inhibited effect of CSIG is coincided well with the experiment results from other system. Mouse CSIG/RSL1D1 was reported to regulate the activity of nucleostemin through modulating its subcellular distribution (in the nucleolus or nucleoplasm) [18]. And nucleostemin was proved to delay the senescence progression in the mice fibroblasts [20]. Thus in the mice fibroblasts, CSIG/ RSL1D1 probably controls the cell aging progression by regulating the activity of nucleostemin (Figure 1).
To study the domain importance of CSIG to regulate cell proliferation and senescence, we performed the domain analysis. The results showed that the ribosomal L1 domain and Lys-rich region were both essential for CSIG to modulate cell proliferation and cellular senescence [21]. We further explored the regulatory mechanism of CSIG expression during senescence. We found that the E2F transcription factor 1 (E2F1) up-regulated CSIG expression by binding and activating CSIG promoter in early-passage cells [22]. While E2F1 expression level and its transcriptional activation effect declined during senescence, which in turn led to the decrease of CSIG expression [22].
CSIG and apoptosis
Mediating the apoptosis pathway under stress is another previously unknown non-ribosomal function for CSIG. Following cell exposure to UV irradiation, p33ING1, a member of inhibitor of growth family member 1 (ING1) family, was accumulated and transferred from nucleus to nucleolus where CSIG bound to and interacted with p33ING1 [6]. CSIG was also induced under the DNA damage stress and influenced the retention time of p33ING1 in nucleolus [6]. By sequestered in nucleolus by CSIG, the nucleolus p33ING1 further triggered apoptosis through activating downstream key molecules such as BCL2 associated X protein (Bax) [6]. Therefore, CSIG is indispensable for p33ING1 to induce apoptosis in respond to UV irradiation.
It is worth mentioning that ribosomal RNA processing 1 homolog B (RRP1B), an interactive protein with CSIG, is reported to regulate E2F1-induced apoptosis [23,24]. Following DNA damage, E2F1 expression increased and firstly activated the transcription of RRP1B, and then RRP1B assisted E2F1 to transcriptionally activate more key molecules to induce apoptosis [24]. As E2F1 regulates other target genes through RRP1B assistance, RRP1B is necessary for E2F1 to induce apoptosis. As the transcriptional targets of E2F1, both CSIG and RRP1B participate in apoptosis process (Figure 1). Although we have known that CSIG and RRP1B promote apoptosis through different pathways, it is still possible that CSIG might interact and cooperate with RRP1B during the process of apoptosis induction through an unknown mechanism so far. And whether CSIG really involves in the E2F1-induced apoptosis needs to be further confirmed (Figure 1).
Perspective
CSIG plays a critical role in not only cellular senescent, but also other processes including cell apoptosis, cell cycle regulation and tumor development (Figure 1). However, many aspects about the function of CSIG and the underlying mechanism still need to be further investigated. Given that cellular senescence is a complicated physiological process regulated by multiple genes and pathways, whether other cell cycle-related proteins and pathways besides PTEN/p27Kip1 are also involved in mediating the influence of CSIG on senescence is worth exploring. Our recent results indicated that cell division cycle 14B (Cdc14B), one of CSIG-regulated genes, is really involved in senescence regulation (unpublished). The analysis of samples from human and mouse suggested that CSIG might well participate in the progression of cell aging in vivo. Therefore, we are constructing a CSIG/RSL1D1 knock-out mouse model to further explore the function and molecular mechanism of CSIG in tissue aging. By confirming the results in vitro, a more solid and comprehensive view on CSIG will be provided for better understanding its roles and the molecular mechanisms which drive its action. On the other hand, further investigation into the fundamental roles of CSIG such as ribosome-associated function will help us to understand the post-transcriptional or translational regulation mechanism of CSIG. Totally, all these studies on CSIG function and the mechanism will contribute to better understanding of the molecular mechanism of cell aging, apoptosis and tumor development, which will in turn provide the experimental basis for the treatment of aging-related disorders and cancer.
Acknowledgments
This work was supported by the National Basic Research Programs of China (Grant No. 2013CB530801).
References
- Packham DK, Alves TP, Dwyer JP, et al. (2012) Relative incidence of ESRD versus cardiovascular mortality in proteinuric type 2 diabetes and nephropathy: results from the DIAMETRIC (Diabetes Mellitus Treatment for Renal Insufficiency Consortium) database. Am J Kidney Dis 59: 75-83.
- Kitada M, Kanasaki K, Koya D (2014) Clinical therapeutic strategies for early stage of diabetic kidney disease. World J Diabetes 5: 342-356.
- Seney FD, Jr, Persson EG, Wright FS (1987) Modification of tubuloglomerular feedback signal by dietary protein. Am J Physiol 252:F83-90.
- Sallstrom J, Carlstrom M, Olerud J, et al. (2010) High-protein-induced glomerular hyperfiltration is independent of the tubuloglomerular feedback mechanism and nitric oxide synthases. Am J Physiol. Regul Integr Comp Physiol 299: R1263-1268.
- Tolins JP, Shultz PJ, Westberg G, Raij L (1995) Renal hemodynamic effects of dietary protein in the rat: role of nitric oxide. J Labo Clin Med 125: 228-236.
- Wen SF, Huang TP, Moorthy AV (1985) Effects of low-protein diet on experimental diabetic nephropathy in the rat. J Labo Clin Med 106: 589-597.
- Dunger A, Berg S, Kloting I, Schmidt S (1997) Functional alterations in the rat kidney induced either by diabetes or high protein diet. Exp Clin Endocrinol Diabetes 105: 48-50.
- Risdon RA, Sloper JC, De Wardener HE (1968) Relationship between renal function and histological changes found in renal-biopsy specimens from patients with persistent glomerular nephritis. Lancet 2: 363-366.
- Gilbert RE, Cooper ME (1999) The tubulointerstitium in progressive diabetic kidney disease: more than an aftermath of glomerular injury? Kidney Int 56: 1627-1637.
- Tang SC, Lai KN (2012) The pathogenic role of the renal proximal tubular cell in diabetic nephropathy. Nephrol Dial Transplant 27: 3049-3056.
- Kitada M, Ogura Y, Suzuki T, Sen S, Lee SM, et al.(2016) Avery-low-protein diet ameliorates advanced diabetic nephropathy through autophagy induction by suppression of the mTORC1 pathway in Wistar fatty rats, an animal model of type 2 diabetes and obesity. Diabetologia. 59:1307-17.
- Kroemer G, Marino G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40: 280-293.
- Archer SL (2013) Mitochondrial dynamics-mitochondrial fission and fusion in human diseases. New Engl J Med 369: 2236-2251.
- Higgins GC, Coughlan MT (2014) Mitochondrial dysfunction and mitophagy: the beginning and end to diabetic nephropathy? Br J Pharmacol 171: 1917-194.
- Hartleben B, Gödel M,Meyer-Schwesinger C, Liu S,Ulrich T,et al. (2010)Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. J Clin Invest 120: 1084-1096.
- Kitada M, Takeda A, Nagai T, Ito H, Kanasaki K, Koya D (2011) Dietary restriction ameliorates diabetic nephropathy through anti-inflammatory effects and regulation of the autophagy via restoration of Sirt1 in diabetic Wistar fatty (fa/fa) rats: a model of type 2 diabetes. Exp Diabetes Res. 908185.
- Yamahara K, Kume S, Koya D, Tanaka Y, Morita Y, et al. (2013) Obesity-mediated autophagy insufficiency exacerbates proteinuria-induced tubulointerstitial lesions. J Am Soc Nephrol 24: 1769-1781.
- Kume S, Uzu T, Horiike K, Chin-Kanasaki M, Isshiki K, et al.(2010) Calorie restriction enhances cell adaptation to hypoxia through Sirt1-dependent mitochondrial autophagy in mouse aged kidney. J Clin Invest 120:1043-1055.
- Bar-Peled L, Sabatini DM (2014) Regulation of mTORC1 by amino acids. Trends Cell Biol 24: 400-406.
- Koya D,Haneda M,Inomata S,Suzuki Y, Suzuki D, et al.(2009) Long-term effect of modification of dietary protein intake on the progression of diabetic nephropathy: a randomized controlled trial. Diabetologia 52: 2037-2045.
- Nezu U, Kamiyama H, Kondo Y, Sakuma M, Morimoto T, et al.(2013) Effect of low-protein diet on kidney function in diabetic nephropathy: meta-analysis of randomised controlled trials. BMJ Open 3 pii: e002934.
- Ideura T, Shimazui M, Morita H, Yoshimura A (2007) Protein intake of more than 0.5 g/kg BW/day is not effective in suppressing the progression of chronic renal failure. ContribNephrol 155: 40-49.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences